The macromolecular engineering group of polymer Lab, taking advantage of its long experience on designing polymer-based responsive self-assemblies, has turned its attention to research activities, targeting potential applications in Biomedicine. In collaboration with Prof. Antimisiaris group of the Department of Pharmacy, they reported in Biomacromolecules a novel complex system, comprising an injectable hydrogel, entrapping liposomes as drug nanocarriers that could be used to tune sustained and controlled drug release.
By using a biocompatible pH-responsive triblock-terpolymer gelator, that form a three dimensional transient network upon switching pH at 7.4 (physiological conditions), they showed that a combination of a physically crosslinked hydrogel with liposomes carrying hydrophilic drugs in their aqueous lumen, can attain prolonged drug release for time periods varying from 14 to 32 days just by adjusting the gelator concentration (1-1.5 wt %). An additional advantage of these kinds of systems is that the drug nanocarriers can be immobilized in a certain tissue (e.g. tumor) due to the high viscosity of the hydrogel medium, which may be proved useful to cancer therapies.
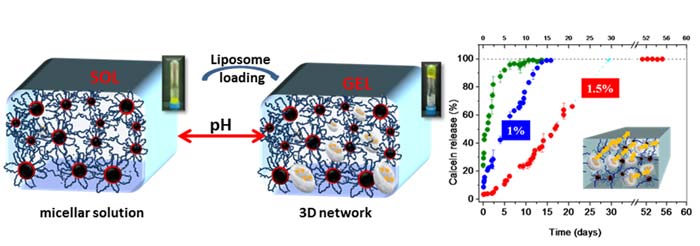
This work received much attention of the Polymer Society since it was incorporated in the top ten most read articles from Biomacromolecules in the full year of 2011. Moreover this work received a poster award in the international conference organized by the Greek Polymer Society, honoring Prof. Hatjichristidis, the October of 2010.
The same group has recently reported in ACS macro Letters a new complex system constituted of biocompatible polymersomes, encapsulating in their aqueous lumen functionalized gold nanoparticles, carrying hydrophobic probe molecules (Nile red, NR) as model drugs. The polymersomes (analogous to liposomes) were formed by self-assembling of a pH-responsive pentablock terpolymer which allows tunability of the hydrophobic membrane permeability by physical (pH) and/or chemical (partial quaternization) ionization of the poly(vinyl pyridine) block. The key feature of this strategy was to combine the advanced properties of polymersomes (i.e. high capacity, tunable membrane permeability, stealth and responsive property), with those of the gold nanoparticles (i.e. easy functionalization, diagnosis capability) in order to create advanced controlled drug delivery systems. It was demonstrated that the highly hydrophobic NR molecules can be delivered from the polymeric nanovehicles in a stepwise fashion, i.e. escape of AuNPs from the polymersome followed by NR release. It is noted that NR cannot escape single-handedly the vesicle wall. This model complex system enables fine tuning of the delivery rate of multiple cargos, of different physico chemical properties, within biological aqueous media of physiological or tumor environmental conditions.
Read more:
dx.doi.org/10.1021/bm2006483 | Biomacromolecules 2011, 12, 3023–3030
dx.doi.org/10.1021/mz300637c | ACS Macro Lett. 2013, 2, 222−225